
-
Whitepaper
Medical Devices
Using Standards to Demonstrate Conformity
Standards provide a voluntary means of demonstrating conformance
The application of standards in the medical devices sector has undoubtedly been accelerated by their use to support regulation.
-
Use of standards to support regulations for medical devices
-
European harmonized standards
-
US FDA recognized standards
-
UK designated standards
-
Standards and the state of the art
-
Monitoring standards development and implementing new or revised standards
BSI is your partner for regulatory compliance
Discover our medical device standards and certification services below:
-
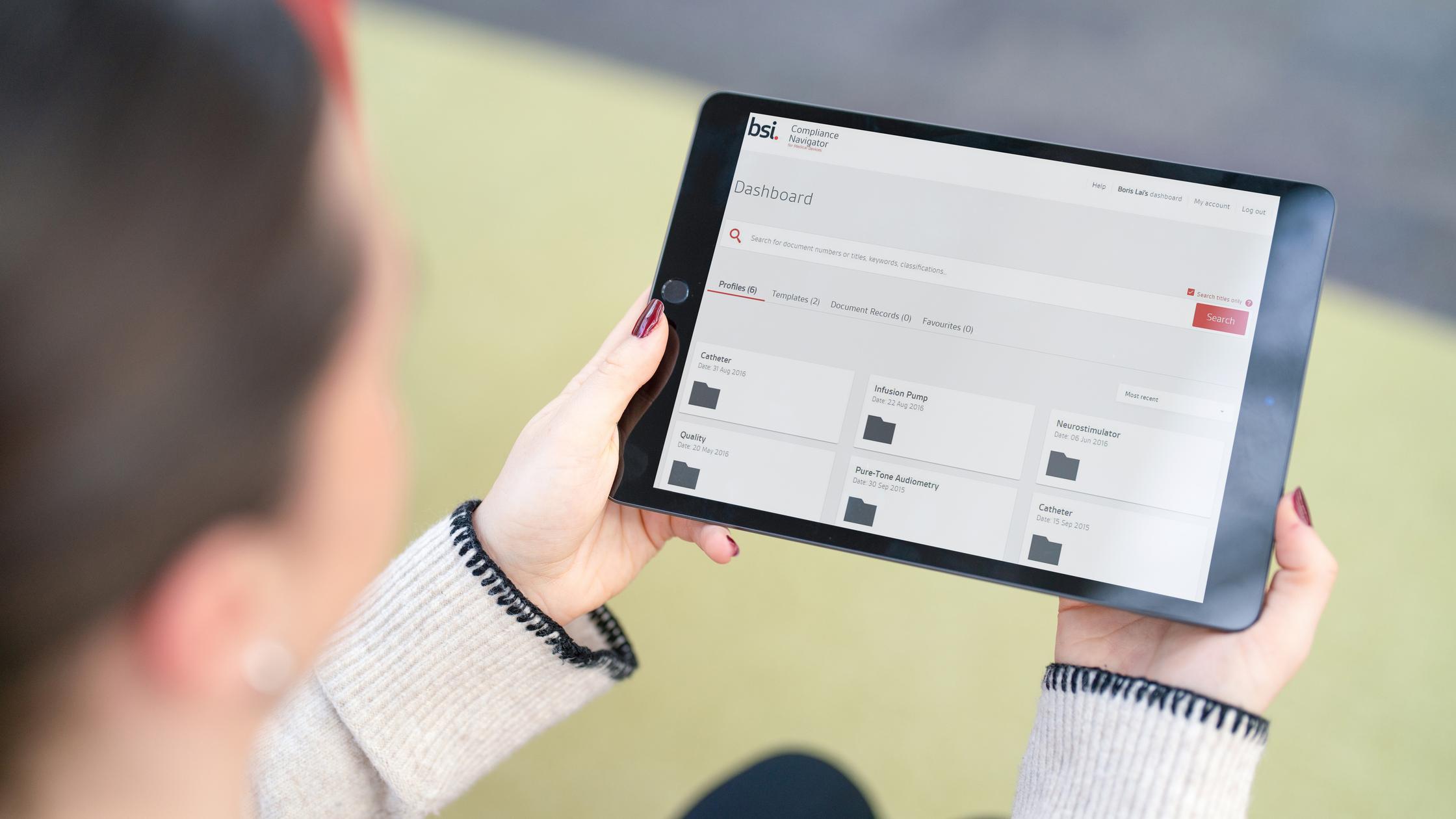
Compliance Navigator
A digital platform transforming how medical device and IVD device manufacturers manage their regulatory documents to save time and reduce risk.
Find out more
-

BSI Knowledge
Deliver quality, improve safety and stay compliant with access to BSI’s healthcare standards. Embed best practice with a company-wide subscription.
Find out more
-
Medical Devices
Our mission is to ensure patient safety whilst supporting timely market access to medical technology in a sustainable manner
Find out more
Speak to the Compliance Navigator team today
Transform your document management process and stay ahead of medtech regulatory updates. Talk with our experts today and arrange a free trial.


